Facilities
Research Infrastructure
The Research Infrastructure has four essential facilities, described as follows:
Facility A) Installation for control of bluefin tuna reproduction. Four tanks with a total volume of around 7,000 m3. Two tanks, 22 and 20 m in diameter and 10 m in depth, intended for breeders, with capacity to hold some 30 tunas each of about 60 kg initial weight and another two of 14 m and 8 m in diameter for juvenile stall. The installation has the possibility of controlling the photoperiod and temperature.
Facility B) Incubation of eggs of bluefin tuna unit. 12 tanks of 500 l of capacity and 50 tanks of 100 l with control of light and temperature.
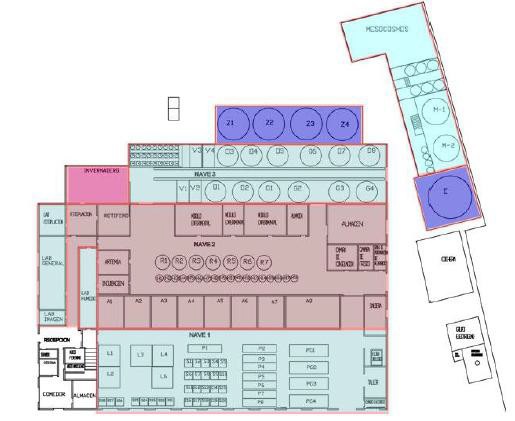
Facility C) Bluefin tuna larval rearing unit, with seven 5,000 liter and eighteen 1,000 liter tanks and two experimental modules with 12 and 27 circular 170 liter tanks. Unit of mesocosmos for the larval rearing consisting of two tanks of 45,000 liters and installation of phytoplankton culture, rotifer, Artemia and copepods, dimensioned to cover the needs of the larval rearing.
All the tanks are provided with water treatment systems, and experimental units are also provided with recirculation systems.
Facility D) Weaning facility ‐ juvenile tuna weaning unit consisting of a cylindrical tank of 100m3, four of 50 m3 and 4 of 20 m with associated recirculation and water treatment system.
Due to great dimensions of the tanks (mainly those devoted to keep broodstock), the four aforementioned facilities (reproduction, incubation, larval rearing and weaning and pre‐ongrowing) are grouped in two close but different building. The first one (building 1) contains the facilities to control reproduction of Atlantic Bluefin Tuna. The other building (building2) contains the facilities for hatching, larval rearing and pre‐ongrowing tuna fry.
Building 1
Facility A, for controlling the reproduction of the bluefin tuna
It consists of a building of 2,660 m2, corresponding 1,960 m2 to the area including the tanks, 300 m2 to the laboratory area and offices and 400 m2 for water recycling and treatment area. The facility count on four big tanks: 2 broodstock tanks (22 y 20 m Ø y 9 m depth 3,500 and 2,500 m3), and 2 juveniles tanks (14 m Ø, 6 m depth, 900 m3 – and 8 m Ø, 3 m depth -150 m3) with a total volume of 7,000 m3. The juvenile tanks are devoted to the quarantine, adaptation and ongrowing of new fish
Each broodstock tank counts on an independent system for the control of the photoperiod and water temperature, and maximum biomass in each one is between 2000 and 3000 kg, which means between 25 and 80 mature tunas. The juveniles’ tanks are designed to keep a higher tuna density and to carry out some ongrowing experiment.
The laboratory and offices area includes a storage room, a cold and freezer room, two laboratories and four small offices. The treatment and recycling water area has two full independent systems including physical and biological filtration, thermal treatment (heating or cooling), chemical filtration with ozone and skimmers, U.V. sterilization and oxygen injection. All the facility is fully equipped with automatic video cameras, activity control of all the devices (pumps, blower, valves, oxygen and ozone machines, UVA equipment, heating and chilling
machines, skimmers, etc.) and sensors of temperature, pH, oxygen, redox potential, measurement of U.V. radiation, photoperiod, flowmeters, water level, etc., which are continuously registering and reporting activities in order to guarantee the correct operation of the systems and to monitor the tuna behavior.
This building is devoted to develop techniques for bluefin tuna reproduction in captivity. This facility is product of a memorandum of understanding signed by the Spanish Ministry of Science and Innovation, the Murcia Regional Community and the IEO, and it is co funded by the regional FEDER funds (80%), but the ownership is only from Spanish Institute of Oceanography. Construction of the building started in 2011 and it was inaugurated in July 2015, and the first fingerlings were allocated in the tanks in August 2015. Another future broodstock has been established in July-August of 2017. In order to obtain eggs meanwhile these fingerlings are growing and they reach their first maturation season, IEO keep a couple of broodstock consisting in 40 big tunas placed in cages along Murcia cost.

Building 2
Facilities for incubation and hatching eggs (B), larval rearing (C), weaning and ongrowing
trials (D) with Atlantic bluefin tuna fingerlings
These facilities extend over a total surface of 8,000 m2 with a building of 3,500 m2. It includes more than 200 culture units, between 200 l and 100 m3 each, with a total volume of 1,300 m3. It provides automatic systems for controlling biological and physic-chemical parameters, as well as monitoring and control systems for feeding, nutrition and fish behavior by means of self-feeders and video cameras.
- Office area (upper floor) of 700 m2.
- Laboratories: Wet laboratory, Laboratories for histology, nutrition, chemistry and biochemistry, lyophilized and sample treatments, Image and video laboratory, Room for feeding preparation and Genetics laboratory
- Water inlet system including 3 submersible pumps of 12.5 kw each (individual water flow 40-60 l/s) and a 15 Kw self-priming emergency pump (water flow of 40-60 l/s).
- Cold and freeze rooms at 2ºC (18 m2) and at –20ºC (27 m2).
- Gas oil heaters of 300 kw each for warmed sea water supply. In close circuit system is able to keep warm a water flow of 40-50 l/s. In open circuit the system is able to heat a water flow of 5 l/s. They are located in an isolated 27 m2 room.
- Sea water cooler system of 43 Kw of cooling performance.
- Biological filter of 36 m3 (3mx3mx4m) in a 28m2 area.
- Air blower system consisting in 6 electrical air blowers of 8 Kw (0.6 – 0.2 Kg/cm2 of pressure).
- Power generator of 222 kw and 250 kVA of electrical power.
- Phytoplankton production unit (Tetraselmis, Nannochloropsis, Isochrysis and Rhodomonas).
- Small isotherm room (30 m2) for phytoplankton strains conservation.
- Small isotherm room (26 m2) for phytoplankton production.
- Greenhouse (80 m2) for massive phytoplankton production.
- Isotherm room for rotifer production and enrichment (29m2) with 4 (1.6 m3) and 5 (600 liters) cylindrical tanks.
- Unit for Artemia hatching and enrichment. Isotherm room (24 m2) with 11 cylinder-conical (160 l) tanks for Artemia hatching and enrichment.
- Unit for copepods (Acartia tonsa) production with 4 (5 m3) broodstock tanks and 8 (1.5 m3) tanks for hatching and growing Nauplii.
- Incubation room (22 m2) provided with 4 (500 l) and 3 (1000 l) cylinder-conical tanks and a small incubation unit provided with 24 (10 l) cylinder-conical tanks. With several extra tanks with small recipients to get a perfect control of incubation temperature.
- Unit for tunids juveniles stockage and preongrowing trials consisting in 1 (100 m3) and 4 (50m3) cylindrical tanks.
- Mesocosm unit (400 m2) for larval rearing consisting in 2 (45 m3) cylindrical tanks and living prey culture facilities: phytoplankton, Rotifer, Artemia and copepod (Acartia) dimensioned for covering the total mesocosm needs
- Building with 4 (23 m3) and 8 (13 m3) cylindrical and 4 (8m3) rectangular tanks to weaning purposes.
- Two water recycled full control unit for larval rearing with 27 (500 l) and 12 (1000 l) cylindrical tanks.
- Greenhouse for phytoplankton massive culture (83 m2).
- Larval rearing and weaning area with 7 (5 m3) and 18 (1m3) cylindrical tanks.
- Two experimental isotherm rooms with a total of 40 (170 l) cylindrical tanks.
- Broodstock area with 7 (45 m3) square-based tanks and 1 (90 m3) rectangular-based tank. This area is mainly devoted to produce yolk sac larvae to supply the tuna larvae needs.
- Weaning and pre-ongrowing area with 4 (23 m3) and 8 (13 m3) cylindrical tanks, 4 (13 m3) and 28 (2 m3) square-based tanks and 8 (7m3) and 4 (14m3) rectangular tanks.
- Room to prepare feeds and enrichers.
- Room to pathology: A small room with 200 l. tanks fully isolated to carry out challenges with pathogens.
Laboratories
- Wet laboratory, fully equipped with microscopes, stereo- microscopes, etc. to observe, analyze, measure and weight phytoplankton, zooplankton, fish eggs and larvae.
- Cold and frozen storage rooms and devices to prepare food.
- Material to samples collection and conservation: freeze-drier, deep-freezer (-80ºC), dry ice.
- Image analyze equipment with Leica programs.
- Heater, stove for culture, isolation of microorganisms.
- Basic nutrition laboratory to determine dry weight, ash, protein and lipid content.
- Molecular biology laboratory provided with:
- Light microscopy facilities formed by a fume hood, a tissue embedding center, a paraffin dispenser, cooler and warming plates, manual and automatic thin-microtome with paraffin or resin cassettes holders.
- The cellular and molecular biology laboratory has a fume hood, a spectral scanning microplate spectrophotometer, a cube spectrophotometer, three microcentrifuges, a termocycler with an additional in situ PCR block, an orbital shaker incubator, a microwave, agatose and acrilamide-bisacrilamode electrophoresis system, white and UV light transilluminators with gel documentation and imaging system, a table-top UV surface sterilizer and a freezer.
- A system to monitor and control different biological and physical-chemical parameters: oxygen, temperature, photoperiod, automatic feeding and behavior control by video cameras.